Press Releases

March 30, 2022
Olympic Ophthalmics Announces FDA Clearance and Availability of Its Next-Generation iTEAR®100 Neurostimulation Device, Expanded Patent Portfolio, and Further Clinical Studies

Jun 06, 2021
Arctic Vision Announces Exclusive Licensing Agreement with Olympic Ophthalmics for iTEAR®100 for the Treatment of Dry Eye Disease in Greater China, South Korea and ASEAN Countries.

April 22, 2021
Olympic Ophthalmics Announces Issuance of Two Seminal Patents for its Neuromodulation Technology.
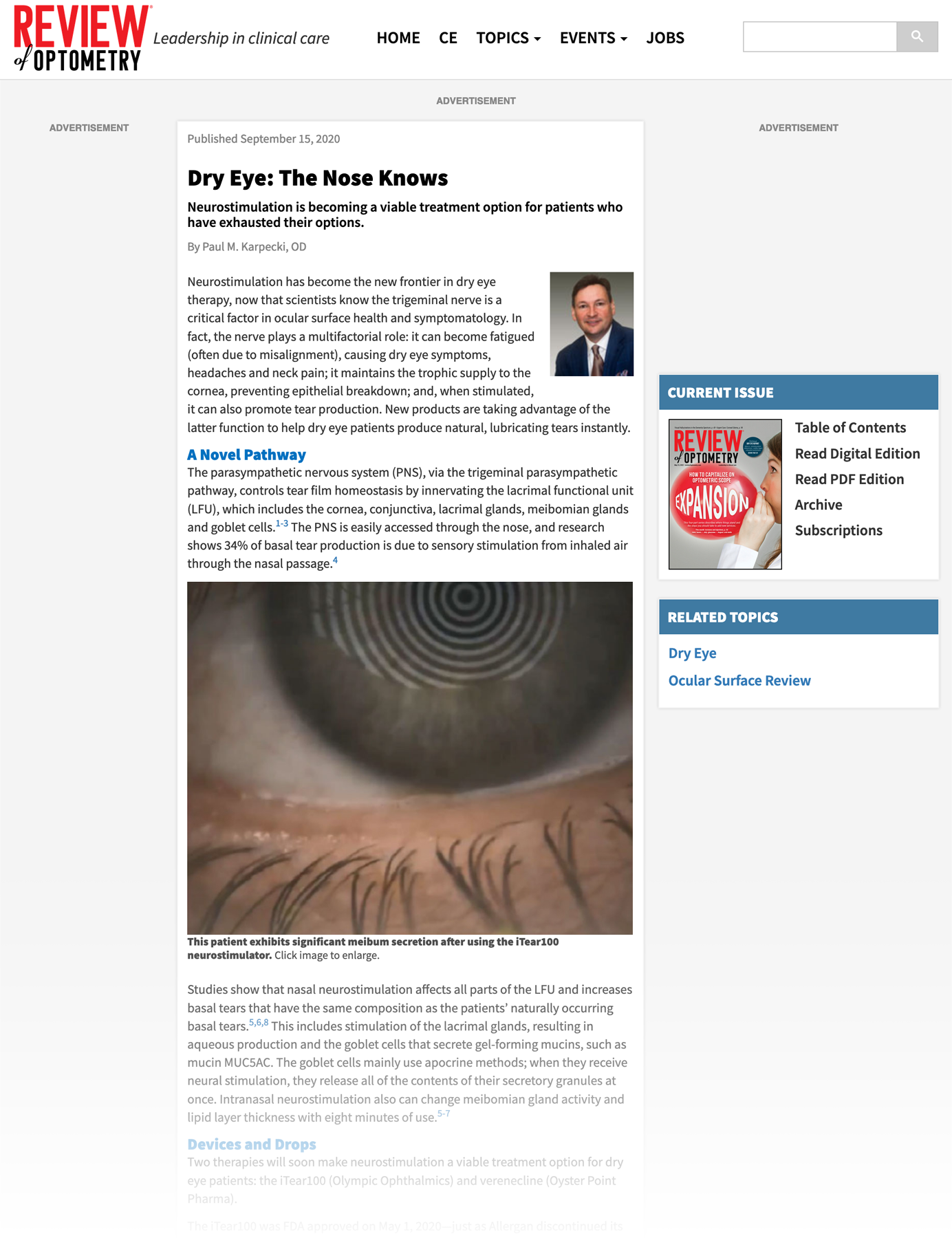
September 15, 2020
Dry Eye: The Nose Knows
Learn how neurostimulation is becoming a viable treatment option for patients who have exhausted their options.
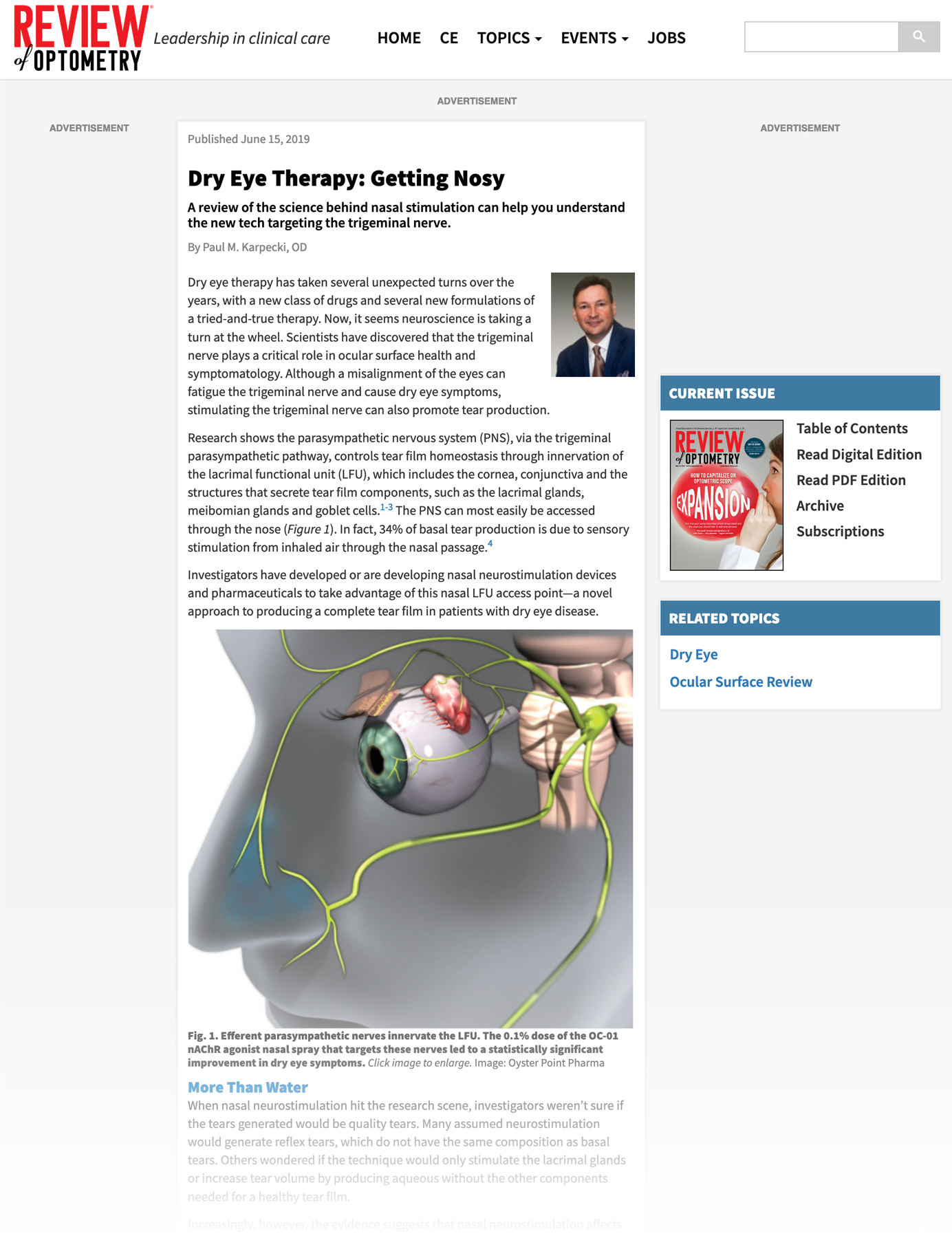
June 15, 2019
Dry Eye Therapy: Getting Nosy
Understand the science behind nasal stimulation.